“Aluminum Potassium Sulphate 500gm” has been added to your cart. View cart

Ascorbic Acid 100gm
$2,500.00 Original price was: $2,500.00.$2,300.00Current price is: $2,300.00.
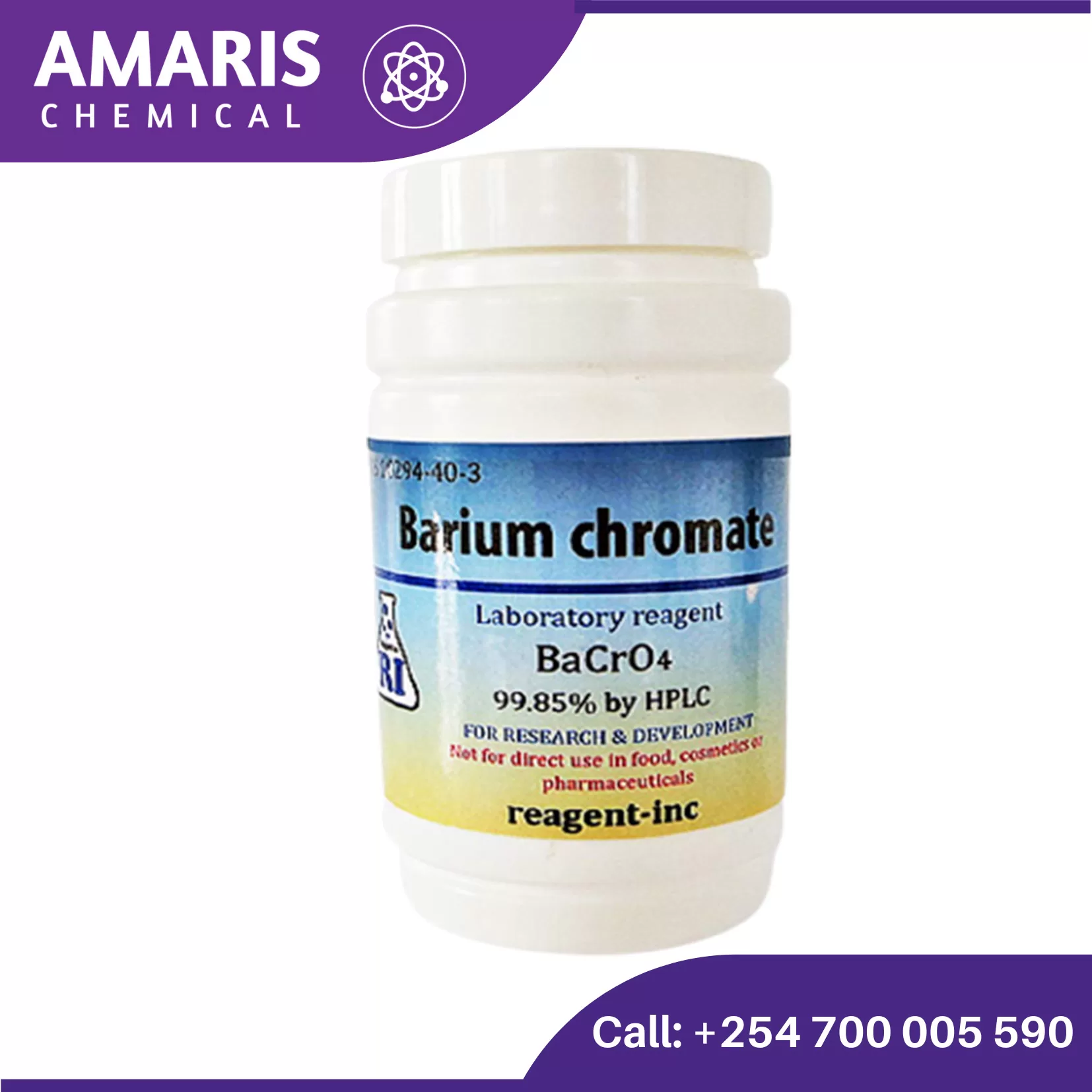
Barium Chromate 250 gm
$2,500.00 Original price was: $2,500.00.$2,300.00Current price is: $2,300.00.
Barium Acetate 500gm
$2,500.00 Original price was: $2,500.00.$2,300.00Current price is: $2,300.00.
SKU:
ACS22783CHEM0
Category: Analytical Reagents
Reviews (0)
Be the first to review “Barium Acetate 500gm” Cancel reply
Shipping & Delivery


MAECENAS IACULIS
Vestibulum curae torquent diam diam commodo parturient penatibus nunc dui adipiscing convallis bulum parturient suspendisse parturient a.Parturient in parturient scelerisque nibh lectus quam a natoque adipiscing a vestibulum hendrerit et pharetra fames nunc natoque dui.
ADIPISCING CONVALLIS BULUM
- Vestibulum penatibus nunc dui adipiscing convallis bulum parturient suspendisse.
- Abitur parturient praesent lectus quam a natoque adipiscing a vestibulum hendre.
- Diam parturient dictumst parturient scelerisque nibh lectus.
Scelerisque adipiscing bibendum sem vestibulum et in a a a purus lectus faucibus lobortis tincidunt purus lectus nisl class eros.Condimentum a et ullamcorper dictumst mus et tristique elementum nam inceptos hac parturient scelerisque vestibulum amet elit ut volutpat.
You may also like…
Anhydrous Barium Chloride 500gm
Barium Chromate 250 gm
Barium Hydroxide 8 hydrate
Rated 5.00 out of 5
Barium hydroxide, with the chemical formula Ba(OH)2text{Ba(OH)}_2Ba(OH)2, is an inorganic compound that is used in various applications. Here’s a detailed overview:
Properties
- Appearance: White crystalline solid.
- Solubility: Moderately soluble in water, forming a strongly alkaline solution.
- Molecular Weight: 171.34 g/mol.
- Density: Approximately 2.18 g/cm³.
- Melting Point: 78 °C (anhydrous form); decomposes in water to form barium oxide and water.
Chemical Behavior
- Basicity: Barium hydroxide is a strong base and dissociates completely in water to form barium ions (Ba2+text{Ba}^{2+}Ba2+) and hydroxide ions (OH−text{OH}^-OH−).
- Reaction with Acids: Reacts with acids to form barium salts and water. For example: Ba(OH)2+2HCl→BaCl2+2H2Otext{Ba(OH)}_2 + 2text{HCl} rightarrow text{BaCl}_2 + 2text{H}_2text{O}Ba(OH)2+2HCl→BaCl2+2H2O
- Hydrate Forms: Often found as the octahydrate (Ba(OH)2⋅8H2Otext{Ba(OH)}_2 cdot 8text{H}_2text{O}Ba(OH)2⋅8H2O).
Barium Nitrate 500gm
Barium Sulphate 500g Extra pure
Barium sulphate is an inorganic compound with the chemical formula BaSO4. It is a white crystalline solid that is insoluble in water, non-toxic, and chemically inert. Barium sulfate is commonly used in various industries and applications, including medical imaging (as a contrast agent for X-ray and CT scans), in paints, coatings, plastics, rubber, and as a filler in various products due to its high density and opacity. Barium sulfate is a white, odorless, and non-toxic crystalline compound with the chemical formula BaSO4, widely used in medical imaging and various industrial applications for its insolubility in water and inert properties.
Related products
2 Propanol 2.5 litres (IPA)
Isopropyl alcohol (IPA), also known as 2-propanol, is a colorless, flammable chemical compound with the molecular formula C3H8O. It is a type of alcohol that is commonly used as a solvent, disinfectant, and cleaning agent in a variety of industries, including healthcare, manufacturing, and electronics. IPA is often used as a disinfectant because it is effective at killing a wide range of microorganisms, including bacteria, viruses, and fungi. It is also commonly used as a solvent for oils, resins, and gums, and as a cleaning agent for electronic devices and other equipment.
Aluminum Carbonate 250g
Aluminum carbonate is a chemical compound with the formula Al2(CO3)3. It is a white, crystalline solid that is insoluble in water. Aluminum carbonate is not commonly encountered in pure form due to its high instability, especially in the presence of water and carbon dioxide. Instead, it tends to decompose into aluminum hydroxide and carbon dioxide when exposed to moisture or acidic conditions.
Aluminum Potassium Sulphate 500gm
Aluminum Potassium Sulphate, commonly known as potassium alum or potash alum, is a chemical compound with the formula KAl(SO₄)₂·12H₂O. It is a type of alum, a double sulfate salt, and is typically found in its dodecahydrate form. Here are some key points about this compound:
Physical Properties:
- Appearance: Colorless, transparent crystals or white powder.
- Solubility: Soluble in water but insoluble in alcohol.
- Melting Point: Decomposes at high temperatures before melting.
Chemical Properties:
- Molecular Formula: KAl(SO₄)₂·12H₂O
- Molecular Weight: 474.39 g/mol (for the dodecahydrate form)
- Acidity: It is slightly acidic in aqueous solution.
Aluminum Sulphate 500gm
Aluminum sulfate, also known as alum, is a chemical compound with the formula Al2(SO4)3. It's commonly used in water treatment plants as a coagulant to clarify turbid or muddy water by causing suspended particles to clump together and settle out. In addition to water treatment, aluminum sulfate has various other industrial applications, such as in paper manufacturing, dyeing, and as a mordant in textile dyeing to help fix dyes to fabrics.
Ammonia Solution 2.5litres
An ammonia solution is a solution of ammonia (NH3) gas dissolved in water. It is a clear, colorless liquid with a pungent odor and a basic pH. The concentration of ammonia in the solution can vary, and is typically expressed in terms of percent by weight or by volume.
Ammonia solutions are commonly used in a variety of applications, including cleaning agents, fertilizers, and as a precursor to other chemicals. They are also used in industrial processes such as refrigeration, gas purification, and water treatment. Ammonia solutions can be dangerous if not handled properly, as they are highly corrosive and can release toxic fumes if mixed with certain chemicals
Ammonium Cupric Chloride
Ammonium Ferric Sulphate 500gm
Sodium Thiosulphate 25kg
Sodium thiosulfate (Na2S2O3) is an inorganic compound that is commonly used as a photographic fixer, as well as in medical and industrial applications. It is a white crystalline powder that is soluble in water and has a mild odor. In photography, sodium thiosulfate is used to remove unexposed silver halide from photographic prints and negatives, making the image permanent. In medicine, it is used as an antidote for cyanide poisoning, and in industrial applications, it is used as a reducing agent, a dechlorinating agent, and in water treatment processes.






























Reviews
There are no reviews yet.